It is very interesting to see how often autism research is noted in the list below.
Item 3: Ricardo Dolmetsch’s work using stem cells to study autism and Timothy syndrome. (discussed here on Left Brain/Right Brain)
Item 10: Public Private Partnerships.
At NIMH and in our broad research community, this has been a year of exciting discoveries and scientific progress, as we strive to make a difference for those with mental illness. Here are 10 breakthroughs and events of 2011 that are changing the landscape of mental health research.
1. Complexity: Discovering New Sources of Genetic Variance.
The discovery of two new sources of genetic variation may have an enormous impact on mental health research.
Students in “Genetics 101” learn that messenger RNA precisely mirrors the DNA sequence from which it was transcribed. However, recent studies suggest a far more complex transmission of information. NIMH-funded researchers compared corresponding RNA and DNA sequences in 27 individuals, and found more than 10,000 sequence sites where the RNA and DNA of the same individual did not match (1). These RNA-DNA mismatches were found in multiple study participants and in different types of cells, including brain and skin cells.
Another study presents what may be the most extraordinary discovery of 2011: somatic ‘retrotransposition’ can alter brain tissue (2). Retrotransposons are mobile genetic elements that can copy and insert themselves within a genome causing mutations in dividing cells. Although these insertions rarely lead to harmful effects when they occur in germ line cells (sperm and egg), they are frequently harmful if they occur in somatic cells, such as neurons. While nearly all studies of the genetics of mental illness have focused on germ line DNA, this new discovery suggests that DNA variation occurring in the developing brain could contribute to mental illness, just as mutations in mature tissues contribute to cancer. These surprising findings suggest a whole new frontier for the biology of mental illness.
2. Transcriptome: Developing Brains Have Unique Molecular Signatures.
Messenger RNAs, or transcripts, are intermediate products that carry the message from DNA, the genetic blueprint, to create proteins, and ultimately, the many different cell types throughout the brain. Each gene can make several transcripts, which are expressed in patterns unique to each of us. To better understand how these patterns of gene expression influence the developing brain, NIMH supported the first map of how RNA expression changes across the life span through two parallel studies of postmortem brains, ranging in age from two weeks after conception to 80 years old (3, 4). The researchers found that nearly 90% of genes are expressed differently during prenatal development, infancy, and childhood. While each of these stages has a distinct transcriptional identity, the fetal brain looks like a different organ compared to the postnatal brain, with 60% of genes expressed differently and 83% of transcripts processed to make unique proteins. Many of the genetic variations associated with mental illness appear to have a specific effect on the form of the gene expressed uniquely during fetal life.
3. Induced Pluripotent Stem Cells: Disease in a Dish.
In 2011, induced pluripotent stem cells (iPSCs) enabled a new round of findings on anomalies in neurodevelopment underlying disorders of mental health. The technology permits scientists to take adult cells and reprogram them to have the capabilities of stem cells to divide and differentiate into specific cell types. Growing iPSCs from adults with diagnosed disorders permits direct observation in cell culture of how the development of neurons is altered in these disorders from the very earliest stages. Scientists studying cells from patients with Timothy syndrome, a condition in which children often show autism-like symptoms, and Fragile X syndrome, an inherited cause of intellectual disability, found the kinds of changes in developing neurons that would disrupt their ability to form normal neural networks and tissues (5, 6). Strikingly, observations of iPSCs derived from patients with schizophrenia showed changes in neurons at stages that would correspond to very early development, years before symptoms emerge (7, 8). These reprogrammed cells also offer a means of medications testing; in these studies, scientists were able to observe the effects of medications in cells from patients with Timothy syndrome and schizophrenia.
4. De Novo Genetic Variants.
This year scientists looking at families with only one case of autism found that up to eight percent of cases in these families were the result of de novo (unique to the person affected) copy-number variants—stretches of DNA that were either multiplied or truncated (9, 10). Analysis of the gene regions affected by these variants implicated a network of genes involved in the development of synapses and neuronal function (11). Another study, focusing specifically on sequences of DNA that code for protein, yielded other de novo genetic changes in one-case families (12). While providing information on genetic contributors to a significant fraction of sporadic autism cases, the work also reveals gene regions for future investigation and ultimately, information on functional changes underlying autism that will offer clues to therapy.
5. Epigenomics: How Experience Alters Behavior.
In any one individual, patterns of gene expression vary widely among cells, leading to a diversity of cell types and functions, even though the cells all have the same DNA sequence. Epigenetic processes—heritable changes in gene expression that are not related to DNA sequence—help explain this diversity. Research suggests that epigenetics may also be a sort of programming language through which experience can have lasting effects on behavior, not only in an individual over a lifetime, but across generations. This effect was demonstrated in a 2011 study of male mice exposed to social defeat—repeated bullying by another aggressive male (13). The bullied males developed behavior resembling depression, and in subtle ways, so did their offspring. This was true even though contact between mother and bullied father was brief and took place well before the birth of the young, suggesting that epigenetic mechanisms played a role. Understanding the nature of epigenetic changes opens possibilities for therapy; scientists also showed this year that they could reverse the silencing of a gene involved in a rare neurodevelopmental disorder, a proof of concept for interventions targeting epigenetic processes (14).
6. Grand Challenges in Global Mental Health.
Mental, neurological, and substance use (MNS) disorders account for 13% of the global burden of disease, more than cancer and cardiovascular disease (15). The Grand Challenges in Global Mental Health initiative, led and funded by NIMH, assembled the largest ever international Delphi panel—over 400 participants representing work conducted in 60 countries—to determine priorities for research relevant to MNS disorders (16). The initiative convened an international community of research funders, engaged them in the consensus-building process, and has already resulted in a $20 million (Canadian) commitment to fund research targeting one Challenge. To date, the Grand Challenges have served as a resource for organizations and governments as they select policy and mental health services priorities. Moreover, the Grand Challenges come at a time of increasing recognition of the economic costs of mental illness (17) and the importance of including mental health in global health care (18, 19).
7. Precision Medicine.
In most fields of medicine, focusing on clinical symptoms is no longer adequate for diagnosis. In line with the National Academy of Sciences’ call for the development of a new nosology based on multiple levels of analysis across medicine, NIMH continues to advance the Research Domain Criteria (RDoC) project. Aiming to define basic dimensions of functioning, from genes to neural circuits to behaviors, RDoC will cut across traditional disorder definitions and facilitate rapid progress in basic neurobiological and behavioral research. In psychiatry, as in other fields of medicine, such an integrated understanding of the foundations of mental disorders may lead the development of new or more personalized treatments.
8. NDAR.
For those familiar with the National Database for Autism Research (NDAR) and its mission to accelerate discovery in autism research, the naming of this resource as one of the top three HHS Secretary’s picks in the HHSinnovates program this fall was well-deserved recognition. As the largest database of its kind to date, NDAR provides approved users with simultaneous access to an unprecedented amount of autism research data, tools, and related resources, drawing on records directly submitted to NDAR as well as from four partner databases—the Autism Speaks’ Autism Genetic Resource Exchange (AGRE) and Autism Tissue Program, the Kennedy Krieger Institute’s Interactive Autism Network (IAN), and the NIH Pediatric MRI Data Repository. Approved NDAR users will have access to data from the 25,000 research participants represented in NDAR, as well as 2,500 AGRE families and more than 7,500 participants who reported their own information to IAN. In the two years since its launch, NDAR has managed to set a new standard for data sharing and collaborative research, not only for autism, but other fields as well.
9. New Culture of Discovery: Team Science.
In an age when events in one country can inspire and incite action in another, so too has global research become a more interconnected and collaborative community. Last year, we saw this with the 1000 Connectomes project, which collected resting state fMRI maps of the brain from over 1000 people around the world and made these results broadly accessible via the Neuroimaging Informatics Tools and Resources Clearinghouse (NITRC). This year, we saw this cultural shift toward team science when the Psychiatric Genomic Consortium reported on genetic variants associated with bipolar disorder and schizophrenia based on over 100,000 samples collected from 200 scientists in 65 institutions and 19 countries. Moreover, 2011 was the year when “standardization, integration, and data sharing” became a mantra for all science at NIMH, ensuring that results from individual labs could be leveraged by the global scientific community.
10. Public Private Partnerships.
As the pharmaceutical industry withdrew from psychiatric medication research and development this year, several new public-private efforts began to re-define the pathway for discovering new treatments. Arch2POCM, a public-private partnership comprising academic, industry, and regulatory scientists and clinicians, created a “precompetitive” initiative, free of intellectual property, for identifying new medications for schizophrenia and autism (20). One Mind for Research grew out of Patrick Kennedy’s moonshot for the mind, building an umbrella organization for neuroscience research related to all brain diseases. The Critical Path Institute led the way with common data elements for clinical research and new tools to promote data sharing. In addition, the Biomarkers Consortium brought industry, advocates, FDA, and NIH together to define biomarkers for neuropsychiatric diseases.
References
(1) Li M, Wang IX, Li Y, Bruzel A, Richards AL, Toung JM, Cheung VG. Widespread RNA and DNA sequence differences in the human transcriptome. Science. 2011 Jul 1;333(6038):53-8.
(2) Baillie JK, Barnett MW, Upton KR, Gerhardt DJ, Richmond TA, De Sapio F, Brennan PM, Rizzu P, Smith S, Fell M, Talbot RT, Gustincich S, Freeman TC, Mattick JS, Hume DA, Heutink P, Carninci P, Jeddeloh JA, Faulkner GJ. Somatic retrotransposition alters the genetic landscape of the human brain. Nature. 2011 Oct 30;479(7374):534-7.
(3) Colantuoni C, Lipska BK, Ye T, Hyde TM, Tao R, Leek JT, Colantuoni EA, Elkahloun AG, Herman MM, Weinberger DR, Kleinman JE. Temporal dynamics and genetic control of transcription in the human prefrontal cortex. Nature. 2011 Oct 26;478(7370):519-23.
(4) Kang HJ, Kawasawa1YI, Cheng F, Zhu Y, Xu X, Li M, Sousa1 AMM, Pletikos M, Meyer KA, Sedmak G, Guennel G, Shin Y, Johnson MB, Krsnik Z, Fertuzinhos MS, Umlauf S, Lisgo SN, Vortmeyer A, Weinberger DR, Mane S, Hyde TM, Huttner A, Reimers M, Kleinman JE, Šestan N. Spatio-temporal transcriptome of the human brain. Nature. 2011 Oct 26;478(7370):483-9.
(5) Pasca SP, Portmann T, Voineagu I, Yazawa M, Shcheglovitov O, Pasca AM, Cord B, Palmer TD, Chikahisa S, Seiji N, Bernstein JA, Hallmayer J, Geschwind DH, Dolmetsch RE. Using iPS cell-derived neurons to uncover cellular phenotypes associated with Timothy Syndrome. Nature Medicine. 2011 Nov 27;17(12):1657-62.
(6) Sheridan, SD, Theriault, KM, Reis, SA, Zhou, F, Madison, JM, Daheron, L, Loring, JF, Haggarty, SJ Epigenetic characterization of the FMR1 gene and aberrant neurodevelopment in human induced pluripotent stem cell models of fragile X syndrome. PLoS ONE 2011 Oct;6:e26203.
(7) Brennand, KJ, Simone, A, Jou, J, Gelboin-Burkhart, C, Tran, N, Sangar, S, Li, Y, Mu, Y, Chen, G, Yu, D, McCarthy, S, Sebat, J, Gage, FH. Modelling schizophrenia using human induced pluripotent stem cells. Nature. 2011 May 12;473(7346):221-5.
(8) Chiang, C-H, Su, Y, Wen, Z, Yoritomo, N, Ross, CA, Margolis, RL, Song, H, and Ming, G-I. Integration-free induced pluripotent stem cells derived from schizophrenia patients with a DISC1 mutation. Molecular Psychiatry 2011 Apr;16:358-360.
(9) Levy D, Ronemus M, Yamrom B, et al. Rare de novo and transmitted copy?number variation in autistic spectrum disorders. Neuron. 2011 Jun 9;70(5):886?97.
(10) Sanders SJ, Ercan?Sencicek AG, Hus V, et al. Multiple recurrent de novo CNVs, including duplications of the 7q11.23 Williams syndrome region, are strongly associated with autism. Neuron 2011 Jun 9;70(5):863?85.
(11 )Gilman SR, Iossifov I, Levy D, et al. Rare de novo variants associated with autism implicate a large functional network of genes involved in formation and function of synapses. Neuron. 2011 Jun 9;70(5):898?907.
(12) O’Roak BJ, Deriziotis P, Lee C, et al. Exome sequencing in sporadic autism spectrum disorders identified severe de novo mutations. Nat Genet. 2011 Jun;43(6):585?9.
(13) Dietz, D.M., LaPlant, Q., Watts, E.L., Hodes, G.E., Russo, S.J., Feng, J., Oosting, R.S., Vialou, V., and Nestler, E.J. Paternal transmission of stress-induced pathologies. Biological Psychiatry 2011 Sep 1;70:408-414.
(14) Huang, H.-S., Allen, J., Mabb, A., King, I., Miriyala, J., Taylor-Blake, B., Sciaky, N., Dutton, J. Jr., Lee, H.M., Chen, X., Jin, J. Bridges, A., Zylka, M., Roth, B., Philpot, B. Topoisomerase inhibitors unsilence the dormant allele of Ube3a in neurons. Nature. Published online ahead of print December 21, 2011, doi: 10.1038/nature10726.
(15) World Health Organization. The Global Burden of Disease: 2004 Update (WHO, 2008).
(16) Collins PY, Patel V, Joestl SS, March D, Insel TR, Daar AS et al. Grand challenges in global mental health. Nature. 2011 Jul 6;475(7354):27-30.
(17) Bloom DE, Cafiero ET, Jané-Llopis E, Abrahams-Gessel S, Bloom LR, Fathima S, Feigl AB, Gaziano T, Mowafi M, Pandya A, Prettner K, Rosenberg L, Seligman B, Stein A, Weinstein C. The Global Economic Burden of Non-communicable Diseases. Geneva, Switzerland: World Economic Forum, 2011
(18) Eaton J, McCay L, Semrau M, Chatterjee S, Baingana F, Araya R, Ntulo C, Thornicroft G, Saxena S. Scale up of services for mental health in low-income and middle-income countries. Lancet. 2011 Oct 29;378(9802):1592-603.
(19) Raviola G, Becker AE, Farmer P. A global scope for global health–including mental health. Lancet. 2011 Nov 5;378(9803):1613-5.
(20) Norman T, Edwards A, Bountra C, Friend S. The Precompetitive Space: Time to Move the Yardsticks. Science Translational Medicine. 2011 March: 3(76): 76cm10.


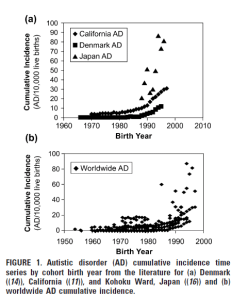
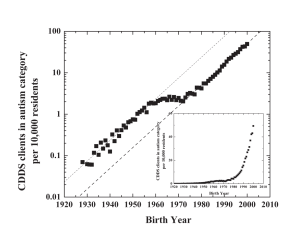
Recent Comments